‘Own your health’ with Hylid Diagnostics
Hylid Diagnostics (HyliDx) will improve the lives of millions of kidney disease patients and save healthcare systems billions of dollars

The problem: A vicious and costly cycle of chronic disease
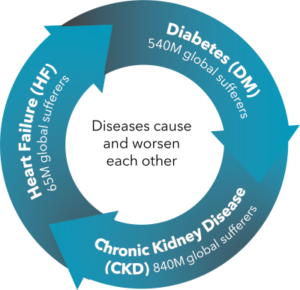
The total US Heathcare cost of the three related diseases in the image—comprising CardioRenal Metabolic Syndrome—is $388 billion
Diabetes costs and outcomes have improved with internet-connected home blood tests and remote care
However, chronic kidney disease (CKD) and heart failure (HF) still suffer from:
- High and growing costs (> diabetes), largely due to unnecessary hospitalization
- Poor outcomes—low quality of life and productivity, high mortality
- No effective home testing or remote care capability
The solution: HyliDx home testing and remote care platform


- Simple, lab-accurate blood tests for chronic disease monitoring
- Test results within 5 min from a small, finger-prick sample of blood
- Results immediately uploaded, via our HyliDx eHealth patient app, to electronic medical records and remote caregivers
- Patients and caregivers can communicate, by messaging or virtual visit, to enhance patient engagement in therapy
- HyliDx Cloud AI flags abnormal data or trends, and eventually predicts and helps avoid complications/hospitalizations
- The first HyliDx home tests are for monitoring chronic kidney disease (CKD)
Our first target: Chronic Kidney Disease (CKD)
Sufferers in US, Canada, and Europe
Total treatment cost
The use: Main applications of the HyliDx system for CKD

Patients with advanced CKD
- To delay onset of costly kidney failure, particularly in remote areas
- 20-25M in North America and Europe
Patients on dialysis
- More than 1M on dialysis in NA, EU
- Dialysis patients need routine monitoring, which is, currently, especially difficult with home dialysis

Doctors' offices
- To save existing CKD patients extra travel to a blood lab prior to their appointments, and to improve early-CKD diagnosis rates (allowing more time for treatment)

High-risk CKD patients at any stage
- For medication dosing/monitoring, and for vulnerable patients on release from hospital to avoid costly readmission
The opportunity
$50B in cost savings
In the US and Canada alone, HyliDx can save healthcare more than $50B in treatment costs

Outstanding team and tech
We have a highly-qualified and experienced senior team (see below), several clinical and business advisors, and a working prototype
Join us early
The company is seeking seed financing to build a customer-ready system
Ready to Make a Difference?
Call us at 613 686 3900 or email us at jack.fairbank@hylidx.com
HyliDx Executive Team
Jack Fairbank, CEO
Past: Senior Key Expert, Siemens Healthineers


Navin Dewagan, CIO
Past: Director of Digital Health and Software, Fresenius Medical


Marius Ivan, Director of Sensor Design
Past: Senior Research Scientist, Philips Research


Sape de Vries, Director of Cartridge Design
Past: Director of Engineering, Siemens Healthineers


Marco Witteveen, Director of Strategy & BD
Past: International Director of Sales & Marketing, Alere


Jack has designed sensors, diagnostic instruments, cartridges, software, and manufacturing systems for 12 years. Recognized as a Senior Key Expert at Siemens, Jack developed a host of critical control systems for next-generation Siemens BGAs, and one of his designs received an innovation award from Popular Scientist. Jack has a PhD from Dalhousie, Postdocs from Harvard and McGill, BEng major and BComm minor from McMaster, Master’s in Business Strategy from Oxford, and a Certificate in Finance from Harvard.
HyliDx Advisors

Seppo Makinen, Business Advisor
12 Exits and over 40 Board Memberships in international Diagnostics and Life Sciences companies

Dr Erika Bariciak, Clinical Advisor
Assistant Professor in the Faculty of Medicine at the University of Ottawa, and Adjunct Professor in the Department of Systems and Computer Engineering at Carleton University

Robert Fraatz, Technical Advisor
Helped lead the development of the OPTI blood analyzer—instrument and sensors—from concept to launch, then acquisition by Roche


Dr Alp Sener, Clinical Advisor (Canada)
Chair and city-wide Chief of Urology and Director of the Kidney/Pancreas Transplant Fellowship Program at the University of Western Ontario

Dr Saif Muhsin, Clinical Advisor (US)
Transplant Nephrologist and Co-Director of Kidney Disease Clinic, Brigham and Women’s Hospital, Harvard University, USA

Dr Amir Bhanji, Clinical Advisor (UK)
Care Group Director for Renal and Transplantation, and Lead for Hemodialysis Program, Portsmouth Hospitals NHS Trust, UK
HyliDx also has contract Regulatory Advisors who are experts in FDA, EU, Health Canada, and UK medical device regulations, as well as ISO 13485 and QMS requirements (for which they are expert auditors)
Contact HyliDx
Hylid Diagnostics Inc. (HyliDx)
2487 Kaladar Avenue, Unit 209, Ottawa, Ontario K1V 8B9 CANADA
Phone: +1 613 686 3900
Email: jack.fairbank@hylidx.com




